Bihar STET Result 2023 Date, BSEB STET Result to be out soon on, Check Update5 hrs ago.UPSC Agriculture Optional Previous Year Question Paper: PDF Download5 hrs ago.NCERT Solutions for Class 12 Accountancy Chapter 2 Reconstitution of Partnership Firm: Admission of a Partner, Download PDF5 hrs ago.Optical Illusion Eye Test: Can you find the hidden sentence in the picture in 9 seconds? 5 hrs ago.Old Parliament vs New Parliament: What Are the Key Differences?5 hrs ago.TS EDCET Counselling 2023 BEd Admission Dates Out, Check Schedule Here5 hrs ago.JEE Main Chemistry Syllabus 2024: Detailed Syllabus with Unitwise Topics, Download PDF5 hrs ago.JEE Main Complete Syllabus 2024: Detailed Syllabus with Unitwise Topics, Download PDF5 hrs ago.JEE Main Mathematics Syllabus 2024: Detailed Syllabus with Unitwise Topics, Download PDF5 hrs ago.Kane Williamson Stats 2023: Total Runs, Centuries and Wickets in All Format 4 hrs ago.Jos Buttler Stats 2023: Total Runs, Centuries and Wickets in All Format4 hrs ago.Brain Teaser to Test Your IQ: Find the missing details in the picture in 10 seconds!3 hrs ago.List of All Kings of Saudi Arabia 3 hrs ago. 
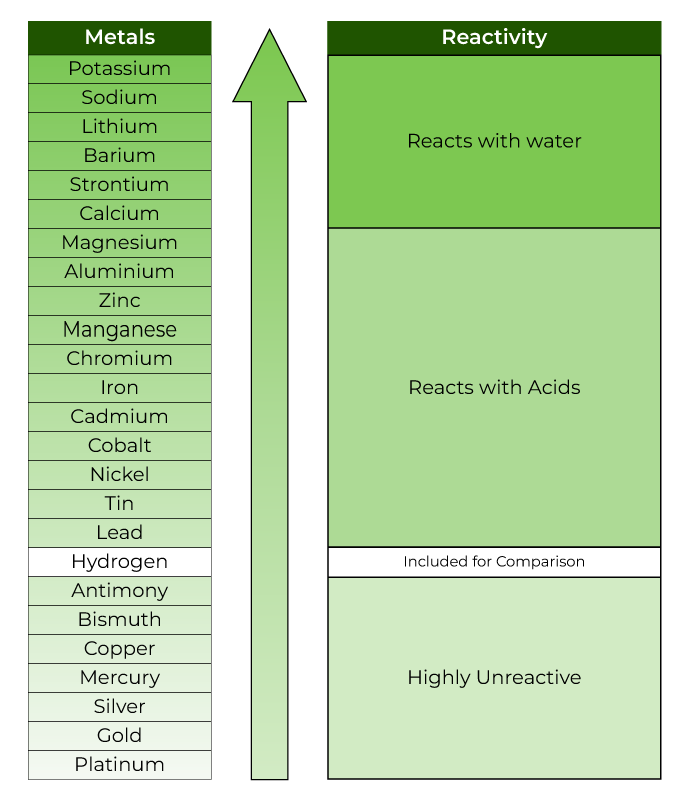
Optical Illusion IQ Test: Only 2% can spot a pizza hidden inside the breakfast table picture in 11 secs!2 hrs ago.School Assembly News Headlines For 19 September: Asian Games 2023, Special Parliament Session, Moto GP India2 hrs ago.In the reactivity series, the most reactive metal is placed at the top whereas the least reactive metal is placed at the bottom. If metal A displaces metal B from its solution, it is more reactive than B. Reaction of Metals with Salt Solutions: Reactive metals can displace any metal less reactive than itself, from the oxide, chloride or sulphate of the less reactive metal in solution or their molten state. Reaction of Metals with Acids: Metals are usually displace hydrogen from dilute acids (Except copper, silver and gold do not displace hydrogen from dilutes acid because they are less reactive than other metals). Reaction of Metals with Water: Metals react with water to form a metal hydroxide and Hydrogen gas. Reaction of Metals with Oxygen: When metal are burnt in air, they react with the oxygen to form metal oxide. The chemical metal chemical properties of metals are discussed below: The electropositive nature allows metals to form compounds with other elements easily. Sodium metal forms sodium ions Na+, Mg forms positively charged Magnesium ions Mg2+and aluminium forms aluminium ions Al3+. Metals are very reactive and tend to losses electrons easily and form positively charged ions therefore metals are called electropositive elements. They have high densities (except Sodium and Potassium).They have high melting and boiling points (except Sodium and potassium).They are solid at room temperature (Except Mercury).Aluminium, Iron, Calcium, Sodium, Potassium, and Magnesium are the major metals is found abundance in the earth’s crust (names are in decreasing order).These elements form positive ions by loosing electrons (or donating).They are good conductor of heat and electricity and are malleable and ductile.Also the elements which are located extremely left have most metallic properties. In the periodic table, these elements are located and confined towards left and middle. There are similarities as well as differences are the properties of these elements on the basis of their, all the element can be divided three types: Metal non- Metal metalloid Metals are those elements which losses electrons and provides cation.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
